Galling occurs when metals adhere to each other during sliding contact. This is especially important when considering threaded connections. If the surfaces adhere during connection, they will be damaged when they are disassembled leading to costly repair or replacement. The ability to disassemble and reassemble a connection is a valuable attribute to many applications. One example where this is especially important is in a drill string. Tubular components in a drill string need to be connected quickly, securely and be assembled rapidly and repetitively.
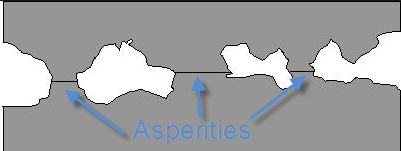
No metal is perfectly smooth. All materials have valleys and ridges forming asperities. The surface areas of these asperities are extremely small creating a large amount of local pressure when they come together, much larger than the overall pressure. Some material combinations are able to form a bond under these conditions. In certain materials, this can be controlled and done purposely to form a strong weld without a heat affected zone. In cases such as threaded connections, this undesirable effect is called galling which leads to thread damage and in extreme cases complete seizure.
Material Interactions In order for galling to occur, there has to be pressure and direct metal to metal contact with heat acting as a catalyst. Different material structures have different tendencies to gall. Some materials have protective oxides that act as a barrier preventing galling. Materials also create different amounts of heat and dissipate that heat at different rates. Another phenomenon is one material might initially gall onto the other with that material acting as a protective layer. What initially seems like a few straight forward factors quickly compounds into an extremely complex interaction.
All these strategies are effective to varying extents but every one of them contains a compromise. The use of lubricating or locking compounds and slowing the speed all negatively affect productivity. Surface hardening steels sacrifice corrosion resistance. Coatings add cost and will have tendency to rub off. Mating two different materials can be effective but there will be an extra set of material properties to consider.